|
||||||
|
||||||||
|
1. Capillary zone electrophoresis (CZE) The capillary zone electrophoresis (CZE) is a new separating method in which molecules are separated by the electro-mobility of anions, cations and neutral molecules to move toward the opposite electrode and the electro-osmotic mobility for cations to move toward the cathode due to the high concentration of cations near the surface of the capillary caused by the cations in the buffer solution that pull the anions on the walls of the capillary. 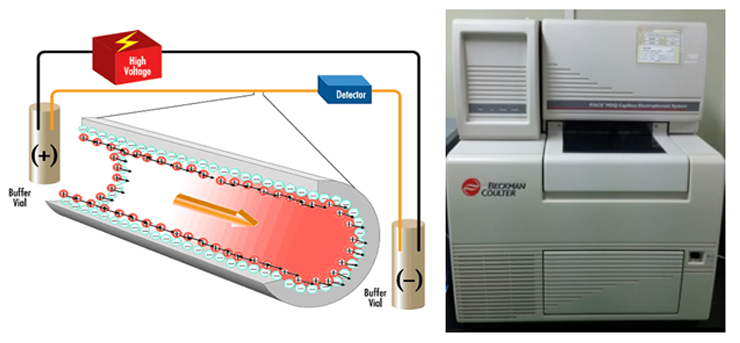 2. Electrophoresis; SDS-PAGE The sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) is a method of separating proteins based on the molecular weights. The basic principle involves physically and chemically denaturizing the proteins to be separated and imposing artificial potentials (-) on them and subsequently letting electric currents flow into certain sizes of pores from (-) ions to (+) ions to separate proteins according to their sizes.The pore sizes in the gel formed by polyacrylamide are adjusted by the ratio between bisacrylamide and acrylamide; the gel is a polymer formed by the cross-linkages of the lateral chain functional bases of acrylamide caused by those compounds with two functional bases such as N,N’-methylenebisacrylamide; effective separation ranges are determined by the ratio between them. In addition, movements of proteins are determined by the moving speeds of ions in the buffer solution based on their sizes. 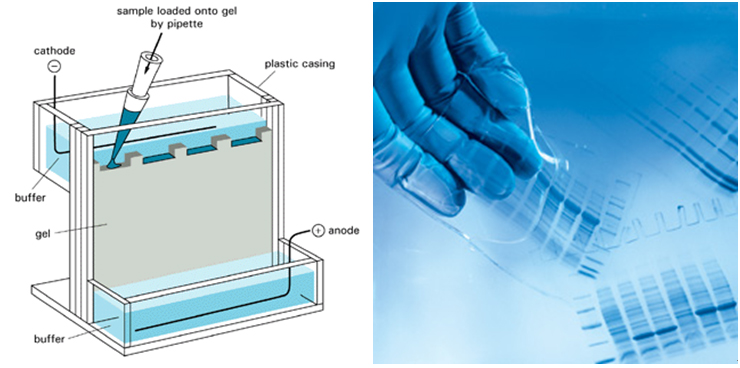 3. Peptide Mapping As methods to elucidate the molecular weights and characteristics of therapeutic proteins, biological or biochemical technologies are being widely used and developed. Of them, peptide mapping enables to digest a target protein using enzymes that digest certain sites in protein structures and separate the digested fragments of it with high performance liquid chromatography (HPLC) in order to analyze the characteristics of them. The chromatography commonly used is reversed phase-HPLC which is commonly used not only to analyze and purify but also to analyze in large quantities of hydrophobic molecules including peptides, proteins, nucleic acids etc. 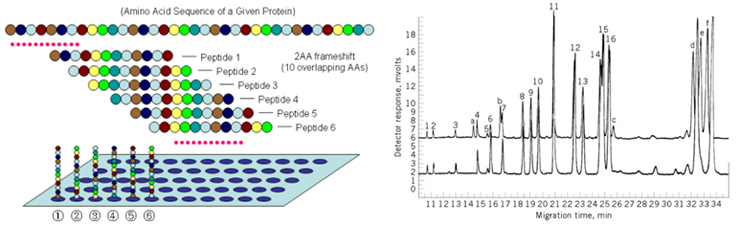 4. Host cell-derived peptide (HCP) The tests to evaluate and predict the safety of it includes approval criteria for residual host cell-derived protein and they are stipulated as approval criteria for production and marketing of therapeutic proteins in foreign countries. If the amount of host cell-derived peptide (HCP) administered into human is more than the suitable level, host cell-derived peptides (host cell-derived peptides and/or protein, HCP) may trigger immunogenicity and thus initiate immune reactions when administered for a long time adversely affecting human. Therefore, the amount of HCP contained in the therapeutic proteins should be analyzed by silver staining, western blot analysis and ELISA (enzyme linked immunosorbent assay) methods as analysis methods. Among them, the ELISA method that uses the specificity of antibody-antigen reactions is widely used. ELISA is known to be an analysis method with excellent and sensitive specificity and selectivity to reactions. Peptides isolated from host cells that produce EPOSIS™ drug substance are used as reference standard. Antibodies against them are obtained and used in EIA method for HCP.  5. Host cell-derived DNA(HCD) To measure the amount of host cell-derived DNA (HCD) existing in drug substance, the HCD is detected using a slot-blotting method and the overall processes can be largely divided into 4 steps. The first one is a step to fix the DNA to be detected in single strands to nylon membranes using the slot-blotting and the next one is a step to synthesize digoxigenin (DIG) labeled DNA probes using a random primed labeling method. Here, the synthesis is done using Klenow enzymes and one DIG-11-dUTP participates per 20~25 bases. The following step is a step of hybridization between the DNA fixed with the slot-blotting method and the DIG-labeled DNA and the final step is a step of immunological detection of the hybridized probes using anti-DIG antibody. In this final stage, the substrate, BCIP/NBT is made to form precipitates by alkaline phosphatase. Here, the degree of color development increases in proportion to the amount of DNA fixed on the membrane.  6. Sialic Acid Content Sialic acid content within these branches are important to determine in vivo half-life of drug substance. Lectin-mediated clearance of glycoprotein in the liver makes glycoproteins that have lower sialic acid contents decomposed. It would be enough to overcome this system that sialic acid contents in drug substance is high. Since sialic acid is reacted with resorcinol to develop blue color proportional to its content, its content in drug substance could be evaluated by measurement of optical density at 580nm.  7. Bacterial endotoxin Limulus Amebocyte Lysate (LAL) is obtained from the blood cells of horseshoe crabsinfected with Limulus polyphemus. LAL is used as a highly sensitive indicator that candetect the existence of the endotoxin of Gram negative bacteria that may be pyrogenicsubstances.LAL interacts with the endotoxin of Gram negative bacteria to show coagulation reactions;the LAL reagent kit commercialized based on such principle can be used to measure theexistence and concentration of endotoxin in the analysis sample. The endotoxin in the analysis sample decomposes and activates the coagulase proenzyme in the LAL reagent. Theactivated coagulase converts the coagulogen into coagulin, which in turn decomposes thecolorless substrate Ac-He-Glu-Ala-Arg-pNA and converts it into yellowish p-nitroaniline(pNA). Add a reaction stop solution to the latter to measure optical density at 405~410 nm.Since the endotoxin content and optical density show linear correlations at concentration of0.1~1 EU/mL, calculate the endotoxin content of the test solution using the standard curve.  8. Western blotting Western blotting is an immunochemical method that involves electrophoresing the protein tobe detected in a membrane matrix and adding an antibody that identifies a particular antigento detect the protein (antigen) bound to the antibody.In other words, this method separates proteins using electrophoresis, transfers them to themembrane (PVDF), and allows them to react with an antibody that can specifically detect erythropoietin. It again allows the proteins to react with a secondary antibody that detectsthe primary antibody; since it can trigger color development, the method enables identifyingthe immunological homogeneity of erythropoietin. 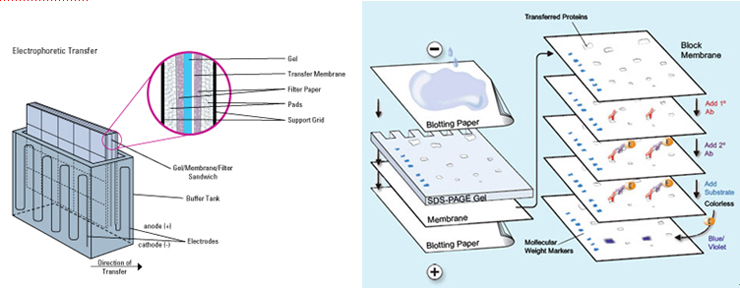 9. Enzyme-linked immunosorbent assay (EIA test) To verify the labeled activity of the final products, identification tests are conductedusing EIA (enzyme-linked immunosorbent assay). As a very sensitive analysis method withexcellent specificity and selectivity to reactions, EIA can measure amounts of even ng orless. The samples to be analyzed are put into the wells to which specifically detectingantibodies are fixed, and antigen-antibody complexes are then formed by antigen-antibodyreactions. To detect these complexes, secondary antibodies linked with enzymes are added to trigger reactions. If substrates of the enzymes are added to these reactants, colors aredeveloped; the degree of color development is then determined by the amount of antigenantibodycomplexes.Obtain the standard curve based on the reference solution and substitute the degree of colordevelopment obtained from the test solution with the standard curve. The protein content ofthe sample can then be measured. 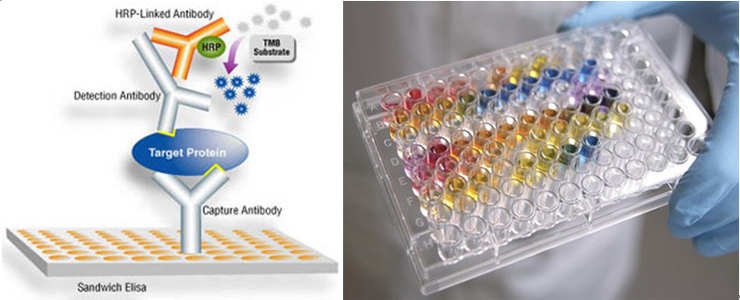 10. High-performance liquid chromatography (HPLC) HPLC is a technique in analytic chemistry used to separate the components in a mixture, to identify each component, and to quantify each component. It relies on pumps to pass a pressurized liquid solvent containing the sample mixture through a column filled with a solid adsorbent material. Each component in the sample interacts slightly differently with the adsorbent material, causing different flow rates for the different components and leading to the separation of the components as they flow out the column.HPLC has been used for medical (e.g. detecting vitamin D levels in blood serum), legal (e.g. detecting performance enhancement drugs in urine), research (e.g. separating the components of a complex biological sample, or of similar synthetic chemicals from each other), and manufacturing (e.g. during the production process of pharmaceutical and biological products) purposes. 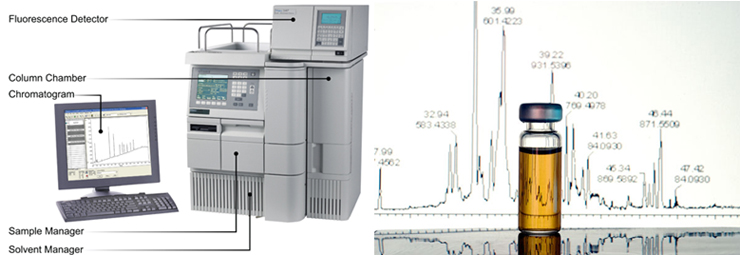 |
 > Service > Analysis
> Service > Analysis

